- Research Article
- Open access
- Published:
Palladium(0) catalyzed Suzuki cross-coupling reaction of 2,5-dibromo-3-methylthiophene: selectivity, characterization, DFT studies and their biological evaluations
Chemistry Central Journal volume 12, Article number: 49 (2018)
Abstract
Thiophene derivatives have shown versatile pharmacological activities. The Suzuki reaction proved a convenient method for C–C bond formations in organic molecules. In the present research work novel derivatives of 2,5-dibromo-3-methylthiophene (3a–k and 3l–p) has been synthesized, via Suzuki coupling reaction in low to moderate yields. A wide range of functional groups were well tolerated in reaction. Density functional theory investigations on all synthesized derivatives (3a–3p) were performed in order to explore the structural properties. The pharmaceutical potential of synthesized compounds was investigated through various bioassays (antioxidant, antibacterial, antiurease activities). The compounds 3l, 3g, 3j, showed excellent antioxidant activity (86.0, 82.0, 81.3%), respectively by scavenging DPPH. Synthesized compounds showed promising antibacterial activity against tested strains. 3b, 3k, 3a, 3d and 3j showed potential antiurease activity with 67.7, 64.2, 58.8, 54.7 and 52.1% inhibition at 50 µg/ml. Results indicated that synthesized molecules could be a potential source of pharmaceutical agents.
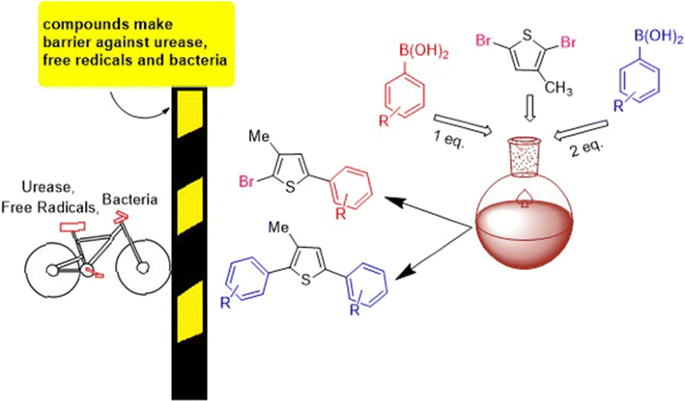
Background
Thiophene is found in central core of various compounds and is well known for its intrinsic electronic properties [1, 2]. A number of thiophene based heterocycles have been reported for versatile pharmacological activities [3,4,5,6,7,8,9]. Biaryl thiophenes are pharmacologically important agents and widely used as anti-inflammatory [10], chemotherapeutic [11], antimicrobial [12] and antioxidant agents [13]. Several reports about regioselective Suzuki coupling of dibromothiophene are available in literature [14, 15]. Palladium catalyzed coupling of 2,5-dibromothiophene has been reported and the yield of obtained product was low (29%) [16]. Synthesis of 2,5-diheteroarylated thiophenes from 2,5-dibromo thiophene derivatives has been reported in good yield [17]. Regioselective Suzuki coupling of 2,5-dibromo-3-hexylthiophene has been reported and preferably coupling occurred at C5 position [18]. The more electron deficient carbon moiety is preferably reactive towards attacking nucleophiles, whereas other reactive carbons do not show any response. Different heterocycles undergo electrophilic substitutions and this regioselectivity can be applied to these substrates [19]. In heterocycles substitution reactions, heteroatom (O, S and N) electron lone pair is being donated to the ring. However, in halogenated thiophenes Suzuki reaction with high oxidative addition, the arylboronate anion preferably attacks the electron deficient carbon bonded with the halogen. And it was observed that transmetallation step is faster due to negatively charged boronate anion then the neutral boronic acids [20]. Extending the scope of Suzuki coupling reaction in regioselective domain a series of 2,5-dibromo-3-methylthiophene derivatives has been synthesized specially with aim to explore their biological importance for the first time.
Results and discussion
Chemistry
A series of thiophene derivatives (3a–k) and (3l–p) has been synthesized by reaction of 2,5-dibromo-3-methylthiophene with variety of arylboronic acids in low to moderate yields (27–63%) (Scheme 1, Table 1).
Synthesis of 2-bromo-3-methyl-5-arylthiophenes (3a–k) and 2,5-diaryl-3-methyl thiophenes (3l–p). Conditions: (i) 1 (128 mg, 0.5 mmol, 1 eq), 2 (0.55 mmol, 1.1 eq), Pd(PPh3)4 (14.5 mg, 2.5 mol%), K3PO4 (212 mg, 1.0 mmol, 2 eq), 1,4-dioxane (2.5 ml), H2O (0.625 ml), 12 h, 90 °C under argon. (ii) 1 (128 mg, 0.5 mmol, 1 eq), 2 (1.25 mmol, 2.5 eq,), Pd(PPh3)4 (34.6 mg, 6 mol%), K3PO4 (424 mg, 2.0 mmol, 4 eq), 1,4-dioxane (2.5 ml), H2O (0.625 ml), 12 h, 90 °C under argon
Under the developed Suzuki reaction conditions, when 1.1 eq of arylboronic acid was used the bromo group at 5 position was selectively substituted and a variety of mono-substituted products was synthesized (3a–k) and double Suzuki cross coupling occurred by using 2.2 eq of arylboronic acids and diaryl derivatives of thiophene were synthesized (3l–p) (Table 1). To increase the substrate scope, the arylboronic acids with both electron donating and withdrawing groups were used. The reaction conditions were tolerant of both electron donating and electron withdrawing arylboronic acids. It was noted that some products were obtained in low yield as 3b, 3h, 3i, 3j, 3k, 3n, 3o which can be attributed to the presence of mixture of mono and di-arylated products in both single and double Suzuki cross coupling reaction and it has been very difficult to separate this reaction mixture and low yields were obtained. This may be due to ineffective transmetallation and reductive elimination in overall reaction cycle [12].
Density functional theory (DFT) studies
DFT investigations were computed by using GAUSSIAN 09 software, in order to explore the structural properties and reactivity’s of synthesized derivatives. First of all, compounds (3a–3p) were optimized by using B3LYP/6-31G(d,p) basis set along with the frequency analysis. After optimization the energy minimized structures were used further for frontier molecular orbitals and molecular electrostatic potential (MEP) analysis on the same basis set.
Frontier molecular orbitals (FMOs) analysis
Nowadays frontier molecular orbitals analysis is well known to explain the reactivity of compounds [21] by using different computational methods. The HOMO/LUMO band gap has direct correlation with the reactivity, e.g. if the band is less the compound will be kinetically less stable (more reactive) and vice versa [22]. The FMOs analysis of all derivatives (3a–3p) was carried out by using B3LYP/6-31G(d,p) basis set. As observed from the HOMO/LUMO, the trend of dispersion of isodensity was almost similar in all compounds. Therefore, as a model here we have given the HOMO/LUMO surfaces of compound 3a only (Fig. 1) (the rest are provided in Additional file 1: Figure S1). The corresponding HOMO and LUMO energies along with band gap are narrated in Table 2.
The isodensity in HOMO of all compounds is dispersed on the benzene and thiophene moieties along with the groups attached to the main skeleton. It is clearly reflected from Fig. 1, that in HOMO orbitals the methyl group attached to the thiophene ring and the groups attached to the para position are directly involved in electronic cloud and electronic transition. Whereas isodensity in LUMO of all compounds reflected the similar trend, the methyl attached to thiophene ring and groups attached to the ortho position of benzene did not participate in electronic cloud. The HOMO–LUMO band gap in all compounds found in the range 3.89–4.67 eV. The smallest band gap observed for 3n i.e. 3.89 eV and largest band gap observed for 3p i.e. 4.67 eV. HOMO–LUMO band gap is reflecting that 3n is most reactive and less stable among all, whereas 3p is most stable and less reactive. This is might be that 3n has more planer structure, due to which transition of electrons is more feasible, whereas in 3p the structure is non-planer and does not facilitating the promotion of electrons to higher orbitals easily.
Molecular electrostatic potential (MEP)
Molecular electrostatic potential study by using quantum chemical tools is useful to explain reactivity, charge separations and monovalent interactions of molecules [23]. ESP analysis of compounds 3a–3p was computed by using DFT/B3LYP/6-31G(d,p) basis and graphics (Fig. 2). The range of MEP values of all compounds are given in Additional file 1: Table S1.
In ESP analysis, the dispersion of electronic density is explained on the basis of different colors e.g. the red color indicates the –ve potential and blue color is indicative of +ve potential [24]. It is cleared from ESP analysis that the electronic density in every compound is dispersed with respect to the electronic effect of group attached to the benzene moiety. The groups attached to the para position of benzene ring have direct effect on the electronic cloud of whole molecule. In 3a, the electron withdrawing group (fluoro) is attached to the benzene ring, due to which the –ve potential is dispersed bromo, chloro and fluoro groups instead of concentrating on benzene ring. Whereas in 3b the –ve potential is concentrated on benzene and thiophene ring due to electron donating effect of –OCH3 attached to the para position on benzene ring. Almost similar kind of effect is observed in ESP analysis of all other synthesized derivatives. If electron donating group is attached to the ortho or para position of benzene moiety the electronic density is concentrated on the benzene and thiophene rings (rather the electronic density also depends on the electron donating ability of group as well), such as in compounds 3c, 3f, 3g, 3h, 3i, 3k, 3m, 3n, 3o and 3p. In all these molecules the –ve potential is concentrated on the benzene and thiophen rings, whereas in the rest of molecules the –ve potential is concentrated on the different groups attached at the different positions of scaffolds (Fig. 2).
Antioxidant activity by DPPH radical scavenging assay
Antioxidants have been broadly studied for their capability to protect cells and organisms from the harm induced by reactive oxidative species (ROS) [25, 26]. So, scientists are more interested to find sources for antioxidants which may be either natural or synthetic.
The DPPH radical has been widely used for determining antioxidant activity of various systems [27]. DPPH radical is purple in colour and antioxidants decay that purple colour of DPPH by capturing free radicals. The potential of DPPH scavenging can be quantified by noting absorbance at 517 nm. A study was designed to determine the antioxidant potential of some novel thiophene derivatives (3a–k and 3l–p), by DPPH radical scavenging assay (Table 3). Ascorbic acid was used as control which exhibited 100% DPPH scavenging at 50 µg/ml. The compounds 3l, 3g, 3j, showed excellent antioxidant activity (86.0, 82.0, and 81.3%), respectively by scavenging DPPH. It is noted that some compounds (3d, 3n) showed mild antioxidant activity with 48.2, 40.9% DPPH radical scavenging at 50 µg/ml. However other compounds showed significant antioxidant activity by scavenging DPPH while some compounds exhibited low activity (Table 3). Mabkhot and coworkers found some thiophene moiety containing compounds inactive towards scavenging DPPH and proved them poor antioxidants [28]. The substituents on ring system have pronounced effect on DPPH radical scavenging [29]. So, in light of this reference, this may be cause of variability in DPPH radical scavenging of thiophene based compounds.
Antibacterial activity
Thiophene and its various derivatives have been reported for potential anti-microbial activity [30,31,32]. To overcome the drug resistance issues it is very important to develop new anti-microbial agents. Generally in the field of pharmaceutical, new drugs are developed by molecular modification of well-known compounds whose activity is already established. So a novel series of thiophene derivatives (3a–k and 3l–p) were screened for anti-bacterial activity against variety of Gram-positive and Gram-negative bacterial strains. Percentage inhibition of bacterial growth was examined at concentration (50 μg/ml). For examining the antibacterial activity of series 3a–k and 3l–p, streptomycin was used as standard drug which showed 100% inhibition against various bacterial strains (Table 4). Compounds 3a, 3k, 3i showed highest activity against P. aeruginosa with % inhibition 67.3, 50.5, 41.1% at 50 μg/ml while compounds 3b, 3h, 3d and 3n showed moderate activity with 39.2, 37.6, 34.9, 20.8% inhibition. This series of thiophene compounds did not show any activity against B. subtilis. When activity was observed against E. coli compounds 3a, 3k, 3i showed excellent activity with 94.5, 72.5, 70.4% inhibition. While 3b, 3h and 3n showed moderate inhibitory effect against E. coli. Compound 3a and 3k showed moderate activity against S. aureus and S. typhimurium while compound 3b and 3i showed low activity against these two strains. It was observed that compounds 3c, 3e, 3f, 3g, 3j, 3l, 3m, 3o and 3p were found inactive against P. aeruginosa, B. subtilis, E. coli, S. aureus and S. typhi (Table 4).
The compounds with both electron donating and withdrawing groups showed good to moderate antibacterial activity. This activity was found promising for future benefits of these compounds as anti-bacterial agents. All the thiophene derivatives that were tested for antibacterial activity were found inactive against B. subtilis. Previous reports about substituents effects on anti-microbial activity of thiophene based compounds are available in literature [31,32,33]. This context is a great deal for researchers to determine the medicinal values of thiophene based compounds.
Antiurease activity
The metalloenzyme urease involved in catalyzing the hydrolysis of urea. It is present in some plant varieties, algae, microbes and as well in soil enzymes [34]. This enzyme is involved in pathogenesis of various diseases and cause significant environmental and agriculture issues [35]. Several compounds have been reported as urease inhibitors to reduce agriculture, environmental, medical issues and to enhance the uptake of urea [36]. Heteroaryl pharmacophores have potential inhibitory activity against bacterial and plant urease [37]. A library novel of thiophene based compounds (3a–k, 3l–p) were screened for antiurease activity (Table 5), where thiourea was used as positive control and it showed 98.3% urease inhibition at 50 µg/ml. From these series of thiophene compounds 3b, 3k, 3a, 3d and 3j showed potential antiurease activity with 67.7, 64.2, 58.8, 54.7 and 52.1% inhibition at 50 µg/ml. It was noted that some compounds 3c, 3e, 3f, 3g, 3h and 3i showed moderate antiurease activity. Some of the novel synthesized products exhibited relatively higher antiurease activity while other products showed moderate urease inhibition effects. It is concluded that compounds with electron donating substituents on aryl ring have pronounced effect on urease inhibition and those compounds showed higher antiurease activity. While compounds with electron withdrawing substituents showed less activity. This may be due to decrease in metal chelating activity caused by electron withdrawing substituents and vice versa. These results are in agreement with previously reported antiurease activity of thiophene based compounds [33,34,35,36,37,38]. According to previous study chelation/removal of nickle ions resulted in inactivation of the enzyme [39]. Therefore change in electronic environment and position and orientation of functional groups can be attributed to variability in antiurease activity of different compounds.
Methods
General
The starting materials were purchased from Fisher Scientific company (Pittsburgh, PA, USA) and Sigma Aldrich Chemical Company (St. Louis MO, USA). Characterization of compounds was done by 1H, 13C NMR Spectra, and melting point determination (for solids). 1H, 13C, NMR Spectra at 500, 126, MHz, respectively. Melting points (°C) were recorded of solid compounds. TLC silica gel plates (0.25 mm) were used for monitoring the reaction. Ultraviolet light (UV) was used for visualization. Spectrometer JMS-HX-110 equipped with a data system was used for recording the EI/MS spectra. For elemental analysis CHNS/O analyzer (Perkin-Elmer 2400 series) was used. Silica gel of various mesh sizes was used (70–230 mesh and 30–400 mesh).
General procedure for synthesis of 3a–k and 3l–p
In a reaction vial stirring bar, catalyst Pd(PPh3)4, 2,5-dibromo-3-methylthiophene (1 eq) was added. A disposable Teflon septum was used to seal vial, which was first evacuated, then purged with argon thrice. 1,4-dioxane solvent was added with syringe with stirring under argon. Stirring of mixture was done at rt for 30 min. After that aryl boronic acid, K3PO4 and water was added [15] and again vial was sealed and purged with argon three times and it was stirred for 12 h at 90 °C, and then cooled to rt. After that, ethyl acetate was used for dilution of mixture, the organic layer was separated and MgSO4 was used for drying this layer and through the vacuum the remaining solvent was evaporated. The purification of crude product was done by the column chromatography by using ethyl-acetate and n-hexane (0–50% gradient) to obtain the desired compounds.
Characterization data
2-Bromo-5-(3-chloro-4-fluorophenyl)-3-methylthiophene (3a)
Obtained as a white solid, mp = 113–114 °C, (86 mg, 56%). 1H NMR (CD3OD, 500 MHz): δ 7.72 (dd, J = 6.5, 2.4 Hz, 1H-aryl), 7.56–7.54 (m, 1H-aryl), 7.33–7.30 (m, 1H-aryl, 1H-thiophene), 1.28 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 110.0, 109.8, 117.0, 121.3, 127.3 (2C), 129.2, 130.5, 141.2, 142.3, 158.9, EI/MS m/z (%): 304.9 [M+H]; 305.5 [M+2, 130.0]; 307.5 [M+4, 31.0]; [M-Me] = 289.0; [M-Me, Br] = 210.5. Anal. Calcd. For C11H7BrClSF: C, 42.14; H, 2.42; Found: C, 42.50; H, 2.68%.
2-Bromo-5-(4-methoxyphenyl)-3-methylthiophene (3b)
Obtained as a brown solid, mp = 98–99 °C, (38 mg, 27%). 1H NMR (CD3OD, 500 MHz): δ 7.45 (d, J = 9.0 Hz, 2H-Aryl), 6.88 (s, 1H-thiophene), 6.92 (d, J = 9.0 Hz, 2H-Aryl), 3.80 (s, 3H-OMe), 2.17 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 12.5, 56.8, 110.8, 115.8 (2C), 126.7, 127.8, 128.5 (2C), 141.5, 143.0, 161.6, EI/MS m/z (%): 284.1 [M+H]; 285.2 [M+2, 90.5]; [M-Me] = 267.2, [M-Br] = 204.2, [M-Br, Me, OMe]+ = 159.0. Anal. Calcd. For C12H11BrOS: C, 49.9, H, 3.92; Found: C, 50.8, H, 3.98%.
2-Bromo-5-(4-chlorophenyl)-3-methylthiophene (3c)
Obtained as a yellow solid, mp = 76–79 °C, (85 mg, 60%). 1H NMR (CD3OD, 500 MHz): δ 7.58 (d, J = 8.7 Hz, 2H-aryl), 7.52 (d, J = 8.7 Hz, 2H-aryl), 7.13 (s, 1H- thiophene), 2.18 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 12.0, 108.4, 127.5, 128.6 (2C), 129.4 (2C), 131.6, 134.2, 140.2, 142.2. EI/MS m/z (%): 288.2 [M+H]; 289.3 [M+2, 130.0]; 291.0 [M+4, 31.8]; [M-Br] = 207.0; [M-Br, Cl fragments] = 172.1. Anal. Calcd. For C11H8BrClS: C, 45.9; H, 2.80; Found: C, 45.0; H, 2.90%.
2-Bromo-5-(3,5-difluorophenyl)-3-methylthiophene (3d)
Obtained as a yellow solid, mp = 78–80 °C, (92 mg, 63%). 1H NMR (CD3OD, 500 MHz): δ 7.21–6.98 (m, 3H-aryl), 6.25 (s, 1H-thiophene), 2.43 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 11.2, 103.5, 109.9 (m), 110.2, 111.2 (2C), 127.9, 136.2, 141.2, 142.3, 165.1 (m). EI/MS m/z (%): 290.0 [M+H]; 291 [M+2, 90.5]; [M-2F] = 250.1, [M-Br] = 209.1, [M-2F, aryl fragments] = 175.0. Anal. Calcd. For C11H7BrF2S: C, 44.28; H, 2.38; Found: C, 44.00; H, 2.42%.
1-(3-(5-Bromo-4-methylthiophene-2-yl)phenyl)ethan-1-one (3e)
Obtained as a brown semisolid, (85 mg, 58%). 1H NMR (CD3OD, 500 MHz): δ 8.08 (d, J = 1.5 Hz, 1H-aryl), 7.98–7.86 (m, 1H aryl), 7.64–7.55 (m, 2H), 7.38 (s, 1H-thiophene), 2.65 (s, 3H-OMe), 2.35 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 12.0, 27.0, 110.6, 126.2, 127.0, 128.6, 129.0, 130.6, 133.7, 137.3, 141.0, 142.5, 197.6. EI/MS m/z (%): 296.0 [M+H]; 297.5 [M+2, 95.3]; [M-MeCO] = 250.9, [M-Br] = 216.1. Anal. Calcd. For C13H11BrOS: C, 51.79; H, 3.76; Found: C, 51.68; H, 4.00%.
2-Bromo-3-methyl-(4-(methylthio)phenyl)thiophene (3f)
Obtained as a white solid, mp = 180–181 °C, (85 mg, 57%). 1H NMR (CD3OD, 500 MHz): δ 7.46 (d, J = 8.5 Hz, 2H-Aryl), 7.25 (d, J = 10.5 Hz, 2H-Aryl), 7.09 (s, 1H-thiophene), 2.48 (s, 3H-SMe), 2.18 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 11.6, 14.8, 110.0, 127.0, 127.3 (2C), 127.7 (2C), 130.1, 139.5, 141.5, 142.0. EI/MS m/z (%): 300.9 [M+H]; 301.9 [M+2, 97.5]; [M-Me] = 283.9, [M-SMe] = 252.6, [M-Br] = 219.0. Anal. Calcd. For C12H11BrS2: C, 47.28; H, 3.82; Found: C, 47.50; H, 3.68%.
2-Bromo-5-(4-iodophenyl)-3-methylthiophene (3g)
Obtained as off white solid, mp = 149–150 °C, (75 mg, 40%). 1H NMR (CD3OD, 500 MHz): δ 7.79 (d, J = 8.7 Hz, 2H-aryl), 7.71 (d, J = 8.7 Hz, 2H-aryl), 6.95 (s, 1H-thiophene), 2.19 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 11.5, 94.0, 110.6, 126.5, 129.0 (2C), 132.5, 138.1, 138.2, 141.2, 144.0. EI/MS m/z (%): 380.0 [M+H]; 381.0 [M+2, 90.7], [M-Br] = 299.0; [M-Br, Me fragments] = 283.7; [M-I, Br fragments] = 172.2. Anal. Calcd. For C11H8BrIS: C, 33.68; H, 2.68; Found: C, 33.57; H, 2.23%.
2-Bromo-3-methyl-5-p-tolylthiophene (3h)
Obtained as a white solid, mp = 110–111 °C, (52 mg, 39%). 1H NMR (CD3OD, 500 MHz): δ 7.89 (d, J = 6.9 Hz, 2H-aryl), 7.58 (d, J = 7.2 Hz, 2H-aryl), 6.98 (s, 1H-thiophene), 2.53 (s, 3H-Me), 2.15 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 11.4, 21.2, 109.9, 125.2 (2C), 127.0, 129.5 (2C), 130.2, 131.9, 141.2, 142.0. EI/MS m/z (%): 268.0 [M+H]; 269.3 [M+2, 96.4]; [M-Br] = 187.0, [M-Me, Br] = 172.0; [M-Br, Me, thiophene] = 91.2. Anal. Calcd. For C12H11BrS: C, 53.89; H, 3.15; Found: C, 54.4; H, 3.28%.
2-Bromo-5-(3,5-dimethylphenyl)-3-methylthiophene (3i)
Obtained as a yellow solid, mp = 120–122 °C, (58 mg, 42%). 1H NMR (CD3OD, 600 MHz): δ 7.21–7.15 (m, 2H-aryl), 7.07 (s, 1H-thiophene), 6.97–6.91 (m, 1H-aryl), 2.33 (s, 6H–2Me), 2.31 (s, 3H–Me); 13C NMR (CD3OD, 150 MHz): δ 11.6, 21.0 (2C), 109.5, 127.4 (3C), 130.6, 133.0, 138.5 (2C), 141.0, 142.6. EI/MS m/z (%): 282.0 [M+H]; 283.0 [M+2, 93.5]; [M-Me] = 264.5; [M-Me, Br fragments] = 186.1. Anal. Calcd. For C13H13BrS: C, 54.5; H, 3.66; Found: C, 54.8; H, 4.23%.
2-Bromo-5-(2,3-dichlorophenyl)-3-methylthiophene (3j)
Obtained as a brown solid, mp = 103–104 °C, (72 mg, 45%). 1H NMR (CD3OD, 600 MHz): δ 7.61–7.58 (m, 1H-aryl), 7.52 (dd, J = 8.0, 1.5 Hz, 1H-aryl), 7.46 (dd, J = 8.0, 1.5 Hz, 1H-aryl), 7.11 (s, 1H-thiophene), 2.20 (s, 3H-Me); 13C NMR (CD3OD, 150 MHz): δ 11.8, 110.4, 127.1 (2C), 127.8, 130.3, 131.0, 133.6, 137.3, 141.3, 142.0. EI/MS m/z (%): 323.0 [M+H]; 324.3 [M+2, 164.3]; 326.0 [M+4, 74.0]; 328.0 [M+6, 10.0]; [M-Br] = 241.0 [M-2Cl, Br fragments] = 171.0. Anal. Calcd. For C11H7BrCl2S: C, 41.0, H, 2.19; Found: C, 41.8, H, 2.42%.
2-Bromo-5-(3-chlorophenyl)-3-methylthiophene (3k)
Obtained as a yellow semisolid, (46 mg, 32%). 1H NMR (CD3OD, 600 MHz): δ 7.63–7.61 (m, 1H-aryl), 7.55–7.52 (m, 2H-aryl), 7.34 (t, J = 7.8 Hz, 1H-aryl), 6.96 (s, 1H-thiophene), 2.19 (s, 3H-Me); 13C NMR (CD3OD, 150 MHz): δ 12.4, 110.4, 124.3, 127.0, 127.8, 128.9, 130.0, 134.0, 135.2, 141.3, 142.0. EI/MS m/z (%): 288.0 [M+H]; 289.3 [M+2, 130.0]; 291.0 [M+4, 31.5]; [M-Me] = 270.3; [M-aryl, Cl fragments] = 174.0. Anal. Calcd. For C11H8BrClS: C, 45.9; H, 2.80; Found: C, 45.3; H, 2.23.
2,5-Bis(3-chloro-4-fluorophenyl)-3-methylthiophene (3l)
Obtained as a yellow solid, mp = 84–86 °C, (100 mg, 56%). 1H NMR (CD3OD, 500 MHz): δ 7.73 (dd, J = 6.6, 2.4 Hz, 2H-aryl), 7.59–7.56 (m, 2H-aryl), 7.27–7.26 (m, 2H-aryl), 7.25 (s, 1H-thiophene), 2.31 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 14.5, 117.0 (2C), 118.5, 121.7 (2C), 126.0, 127.5 (m), 128.5, 129.4 (m), 130.0 (m), 133.2, 134.2, 138.3, 158.5 (m), EI/MS m/z (%): 356.0 [M+H]; 358.0 [M+2, 65.0]; 360.0 [M+4, 10.6]; 319.0 [M-Me, F fragments], 300.0 [(M+4), Me, 2F fragments]. Anal. Calcd. For C17H10Cl2F2S: C, 57.4, H, 2.84; Found: C, 57.0, H, 2.82.
2,5-Bis(4-methoxyphenyl)-3-methylthiophene (3m)
Obtained as a brown solid, mp = 90–91 °C, (90 mg, 58%). 1H NMR (CD3OD, 500 MHz): δ 7.51 (d, J = 9.0, 4H-Aryl), 7.38 (d, J = 9.0, 4H-Aryl), 7.07 (s, 1H-thiophene), 3.81 (s, 6H-OMe), 2.17 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 14.2, 55.2 (2C), 114.0 (4C), 126.0 (2C), 126.4, 128.5 (4C), 133.0, 134.2, 138.0, 160.6 (2C), EI/MS m/z (%): 311.0 [M+H]; 295.2 [M-Me]; 203.4 [M-Aryl, OMe fragments]; Anal. Calcd. For C19H18O2S: C, 73.5, H, 5.84; Found: C, 73.0, H, 5.82.
3-Methyl-2,5-bis(4-(methylthio)phenyl)thiophene (3n)
Obtained as off-white solid, mp = 160–161 °C, (75 mg, 44%). 1H NMR (CD3OD, 500 MHz): δ 7.41 (d, J = 8.0, 4H-Aryl), 7.31 (d, J = 8.5, 4H-Aryl), 7.21 (s, 1H-thiophene), 2.51 (s, 6H-SMe), 2.31 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 14.8 (2C), 15.1, 126.5, 127.4 (4C), 127.6 (4C), 130.0 (2C), 133.0, 134.6, 138.0, 139.4 (2C), EI/MS m/z (%): 343.9 [M+H]; [M-Me]+ = 327.0, [M-Aryl, 2-SMe]+ = 173.0. Anal. Calcd. For C19H18S3: C, 66.6; H, 5.30; Found: C, 66.4; H, 5.70%.
2,5-Bis(3,5-dimethylphenyl)-3-methylthiophene (3o)
Obtained as colorless oil, (45 mg, 29%). 1H NMR (CD3OD, 500 MHz): δ 7.21–6.98 (m, 6H-aryl), 6.94 (s, 1H-thiophene), 2.34 (s, 12H-Me), 2.14 (s, 3H-Me); 13C NMR (CD3OD, 126 MHz): δ 14.2, 21.6 (4C), 126.2, 127.3 (4C), 130.6 (2C), 133.0, 133.8 (2C), 134.0, 138.2, 138.9 (4C), EI/MS m/z (%):307.0 [M+H]; [M-Me]+= 291.0; [M-2Me]+ = 276.0. [M-5Me]+ = 231.0. Anal. Calcd. For C21H22S: C, 82.3, H, 7.24; Found: C, 82.1, H, 7.82.
2,5-Bis(2,3-dichlorophenyl)-3-methylthiophene (3p)
Obtained as brown solid, mp = 110–111 °C, (105 mg, 53%). 1H NMR (CD3OD, 500 MHz): δ 7.52 (dd, J = 7.8, 1.2 Hz, 2H-aryl), 7.47–7.46 (m, 2H-aryl), 7.34–7.30 (m, 2H-aryl), 7.10 (s, 1H-thiophene), 2.20 (s, 3H); 13C NMR (CD3OD, 126 MHz): δ 15.5, 126.4, 127.2 (2C), 127.7 (2C), 130.2 (2C), 131.4 (2C), 133.2, 133.8 (2C), 134.5, 138.3 (3C), EI/MS m/z (%):389.0 [M+H+]; 391.0 [M+2, 131.0]; 393.0 [M+4), 63.9]; 395.0 [M+6, 14.0]; 397.0 [M+8), 1.2]; [M+-2Cl fragments] = 316.0; [M+-3Cl fragments] = 281.0; Anal. Calcd. For C17H10Cl4S: C, 51.6, H, 2.60; Found: C, 51.1, H, 2.82.
Computational methods
By using Gaussian 09 software [40] all simulations were performed and visualization of results was accomplished with Gauss view 05 [41]. All compounds geometries (3a–3p) were optimized by using B3LYP/6-31G(d,p) basis set at DFT level of theory. Frequency calculations at same level of theory proved true optimization (where no imaginary frequency was observed). Frontier molecular orbital (FMOs) analysis and molecular electrostatic potential (MEP) were carried out at same basis set as used for optimization.
Pharmacology
General procedure for antioxidant potential of synthesized compounds by DPPH radical scavenging activity
The DPPH radical scavenging was determined by following the reported method [42]. In the reaction mixture 50 µg/ml of test sample and 1 ml of DPPH (2,2-diphenyl-1-picrylhydrazyl) solution (90 μM) was added and mixture volume was made up to 3 ml. Then incubation of mixture was done at rt for 1 h and absorbance of solution was observed at 515 nm. Sample that contained only methanol was used as blank. Percentage DPPH radical scavenging was calculated by following formula:
where, As = absorbance of sample and Ac = absorbance of control (DPPH solution in methanol without sample).
General procedure for Antiurease activity
Firstly, phosphate buffer (200 µl, ~ pH = 7) having one unit of enzyme followed by addition of phosphate buffer (230 μl) and stock solution (20 μl) (thiourea or test sample). The mixture was shaked well and at 25 °C it was incubated for 5 min. After this, 400 µl of urea stock (20 mM) solution was added in every sample tube. With no urea solution the calibration mixture was prepared and positive control solution was prepared with no thiourea solution. Then prepared sample solutions were incubated at 40 °C (for 10 min). After this the phenol hypochlorite reagent (1150 μl) was added. For formation of complex and colour development the tubes were further incubated for 25 min at 56 °C. After cooling a blue colour complex appeared and absorbance was observed at 625 nm and % inhibition was calculated by the following formula:
The IC50 values were determined using the EZ-fit kinetic data base [43, 44].
General procedure for antibacterial activity
The antibacterial activity of novel molecules was carried out by following already reported method [45] against Gram positive (Staphylococcus aureus, Bacillus subtilis) and Gram negative (Pseudomonas aeruginosa, Escherichia coli, Salmonella typhi, Shigella dysenteriae) strains. The bacterial strains were provided by Agha Khan University of Karachi, Pakistan. Streptomycin (50 µg/ml) was used as the positive control. Activity was determined by 96 well plate method. In every well sterilized broth (175 µl) was added and glycerol stock (5.0 µl) bacterial strain was inoculated. The initial absorbance reading maintained between 0.12 and 0.19 and in an incubator bacteria allowed to grow overnight. After 12 h, test sample (20 µl) was added in wells (sample conc was 20 µl/well). The 96 well plates were further incubated (at 37 °C) for 24 h. After incubation the absorbance at 630 nm was observed by using Elisa reader. The difference in absorbance was used as bacterial growth index. Percentage inhibition of bacterial growth was determined by the following formula:
Conclusion
For the synthesis of some thiophene based pharmaceutically important compounds simple, mild, scalable protocols were developed. The optimized method exhibit enhanced substrate scope and expanded functional group compatibility allowing the synthesis of bundle of novel thiophene based structures in significant yields. Frontier molecular orbitals (FMOs) analysis revealed that 3n is most reactive having HOMO–LUMO band gap 3.89 eV, whereas HOMO–LUMO band gap for 3p found 4.67 eV, and is most stable among all. The MEP investigation provided us the idea about the electro and nucleophilic nature of synthesized compounds, and it was envisaged that dispersion of electronic density is highly dependent on nature of groups attached to the aromatic ring. The compounds were screened for biological activities (antibacterial, antiurease and antioxidant). All the tested compounds showed promising biological activities. In light of this research it is concluded that synthesized thiophene derivatives might be a potential source of therapeutic agents. Future investigations in this dimension will provide new visions towards development of novel pharmaceutically important drugs. And these compounds may also be used as intermediates in preparation of fine chemicals for industrial purposes.
References
Damit EF, Nordin N, Ariffin A, Sulaiman K (2016) Synthesis of novel derivatives of carbazole–thiophene, their electronic properties, and computational studies. J Chem 2016:14
Rizwan K, Zubair M, Rasool N, Ali S, Zahoor AF, Rana UA, Khan SU-D, Shahid M, Zia-Ul-Haq M, Jaafar HZ (2014) Regioselective synthesis of 2-(bromomethyl)-5-aryl-thiophene derivatives via palladium (0) catalyzed Suzuki cross-coupling reactions: as antithrombotic and haemolytically active molecules. Chem Cent J 8:74
Chaudhary A, Jha K, Kumar S (2012) Biological diversity of thiophene: a review. J Adv Sci Res 3:3–10
Mohan C, Bhargava G, Bedi PM (2009) Thieno [3,2-d] pyrimidin-4-one derivatives as potential antibacterial agents. J Life Sci 1:97–101
Folkes AJ, Ahmadi K, Alderton WK, Alix S, Baker SJ, Box G, Chuckowree IS, Clarke PA, Depledge P, Eccles SA, Friedman LS, Hayes A, Hancox TC, Kugendradas A, Lensun L, Moore P, Olivero AG, Pang J, Patel S, Pergl-Wilson GH, Raynaud FI, Robson A, Saghir N, Salphati L, Sohal S, Ultsch MH, Valenti M, Wallweber HJ, Wan NC, Wiesmann C, Workman P, Zhyvoloup A, Zvelebil MJ, Shuttleworth SJ (2008) The identification of 2-(1H-indazol-4-yl)-6-(4-methanesulfonyl-piperazin-1-ylmethyl)-4-morpholin-4-yl-t hieno[3,2-d]pyrimidine (GDC-0941) as a potent, selective, orally bioavailable inhibitor of class I PI3 kinase for the treatment of cancer. J Med Chem 51:5522–5532
Laddha SS, Bhatnagar SP (2009) A new therapeutic approach in Parkinson’s disease: some novel quinazoline derivatives as dual selective phosphodiesterase 1 inhibitors and anti-inflammatory agents. Bioorg Med Chem 17:6796–6802
Alagarsamy V, Raja Solomon V, Meenac R, Ramaseshu K, Thirumurugan K, Murugesan S (2007) Design and synthesis of 2-methylthio-3-substituted-5, 6-dimethylthieno [2, 3-d] pyrimidin-4 (3H)-ones as analgesic, anti-inflammatory and antibacterial agents. Med Chem 3:67–73
Wardakhan W, Abdel-Salam O, Elmegeed G (2008) Screening for antidepressant, sedative and analgesic activities of novel fused thiophene derivatives. Acta Pharm 58:1–14
Ding H, Chen Z, Zhang C, Xin T, Wang Y, Song H, Jiang Y, Chen Y, Xu Y, Tan C (2012) Synthesis and cytotoxic activity of some novel N-pyridinyl-2-(6-phenylimidazo[2,1-b]thiazol-3-yl)acetamide derivatives. Molecules 17:4703–4716
Pillai AD, Rathod PD, Xavier FP, Padh H, Sudarsanam V, Vasu KK (2005) Tetra substituted thiophenes as anti-inflammatory agents: exploitation of analogue-based drug design. Bioorg Med Chem 13:6685–6692
Mohareb RM, Wardakhan WW, Elmegeed GA, Ashour RM (2012) Heterocyclizations of pregnenolone: novel synthesis of thiosemicarbazone, thiophene, thiazole, thieno [2,3-b] pyridine derivatives and their cytotoxicity evaluations. Steroids 77:1560–1569
Elsabee MZ, Ali EA, Mokhtar SM, Eweis M (2011) Synthesis, characterization polymerization and antibacterial properties of novel thiophene substituted acrylamide. React Funct Polym 71:1187–1194
Ferreira IC, Queiroz M-JR, Vilas-Boas M, Estevinho LM, Begouin A, Kirsch G (2006) Evaluation of the antioxidant properties of diarylamines in the benzo [b] thiophene series by free radical scavenging activity and reducing power. Bioorg Med Chem Lett 16:1384–1387
Theeramunkong S, Caldarelli A, Massarotti A, Aprile S, Caprioglio D, Zaninetti R, Teruggi A, Pirali T, Grosa G, Tron GC (2011) Regioselective Suzuki coupling of dihaloheteroaromatic compounds as a rapid strategy to synthesize potent rigid combretastatin analogues. J Med Chem 54:4977–4986
Dang TT, Rasool N, Dang TT, Reinke H, Langer P (2007) Synthesis of tetraarylthiophenes by regioselective Suzuki cross-coupling reactions of tetrabromothiophene. Tetrahedron Lett 48:845–847
Borghese A, Geldhof G, Antoine L (2006) Direct C–H arylation of 3-methoxythiophene catalyzed by Pd. Application to a more efficient synthesis of π-alkoxy-oligothiophene derivatives. Tetrahedron Lett 47:9249–9252
Belkessam F, Mohand A, Soule JF, Elias A, Doucet H (2014) Palladium-catalyzed 2,5-diheteroarylation of 2,5-dibromothiophene derivatives. Beilstein J Org Chem 10:2912–2919
Ikram HM, Rasool N, Ahmad G, Chotana GA, Musharraf SG, Zubair M, Rana UA, Zia-Ul-Haq M, Jaafar HZ (2015) Selective C-arylation of 2,5-dibromo-3-hexylthiophene via Suzuki cross coupling reaction and their pharmacological aspects. Molecules 20:5202–5214
Brandsma L, Vasilevsky SF, Verkruijsse HD (1999) Application of transition metal catalysts in organic synthesis. Springer, Berlin
Amatore C, Bahsoun AA, Jutand A, Meyer G, Ntepe N, Ricard L (2003) Mechanism of the Stille reaction catalyzed by palladium ligated to arsine ligand: PhPdI (AsPh3)(DMF) is the species reacting with vinylstannane in DMF. J Am Chem Soc 125:4212–4222
Ahmed MN, Yasin KA, Ayub K, Mahmood T, Tahir MN, Khan BA, Hafeez M, Ahmed M (2016) Click one pot synthesis, spectral analyses, crystal structures, DFT studies and brine shrimp cytotoxicity assay of two newly synthesized 1,4,5-trisubstituted 1,2,3-triazoles. J Mol Struct 1106:430–439
Arshad MN, Asiri AM, Alamry KA, Mahmood T, Gilani MA, Ayub K, Birinji AS (2015) Synthesis, crystal structure, spectroscopic and density functional theory (DFT) study of N-[3-anthracen-9-yl-1-(4-bromo-phenyl)-allylidene]-N-benzenesulfonohydrazine. Spectrochim Acta Part A Mol Biomol Spectrosc 142:364–374
Arshad MN, Bibi A, Mahmood T, Asiri AM, Ayub K (2015) Synthesis, crystal structures and spectroscopic properties of triazine-based hydrazone derivatives; a comparative experimental–theoretical study. Molecules 20:5851–5874
Ahmed MN, Yasin KA, Mahmood T, Wasim F, Khan MH, Tahir MN, Zafar S, Anjum S (2015) Synthesis and structural investigations of new 4-hexyl-1-(4-nitrophenyl)-1-H-1, 2, 3-triazole: an experimental and theoretical insight. Chin J Struct Chem 34:1830–1840
Borut P, Fink R (2014) The protective role of antioxidants in the defence against ROS/RNS-mediated environmental pollution. Oxid Med Cell Longev 2014:22
Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O (2012) Oxidative stress and antioxidant defense. World Allergy Organ J 5:9–19
Can Z, Dincer B, Sahin H, Baltas N, Yildiz O, Kolayli S (2014) Polyphenol oxidase activity and antioxidant properties of Yomra apple (Malus communis L.) from Turkey. J Enzyme Inhib Med Chem 29:829–835
Mabkhot YN, Barakat A, Al-Majid AM, Alshahrani S, Yousuf S, Choudhary MI (2013) Synthesis, reactions and biological activity of some new bis-heterocyclic ring compounds containing sulphur atom. Chem Cent J 7:112–120
Hwang K-J, Kim H-S, Han I-C, Kim B-T (2012) Synthesis of heterocyclic chalcone derivatives and their radical scavenging ability toward 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radicals. Bull Korean Chem Soc 33:2585–2591
Badiceanu CD, Larion C (2009) Antimicrobial activity of some new thioureides from 2-thiopheneacetic acid. Farmacia 57:771–779
Srivastava S, Das B (2011) Synthesis and evaluation of some novel thiophenes as potential antibacterial and mycolytic agents. Der Pharma Chemica 3:103–111
Nasr T, Bondock S, Eid S (2014) Design, synthesis, antimicrobial evaluation and molecular docking studies of some new thiophene, pyrazole and pyridone derivatives bearing sulfisoxazole moiety. Eur J Med Chem 84:491–504
Noreen M, Rasool N, Gull Y, F-u-H Nasim, Zahoor AF, Yaqoob A, Kousar S, Zubair M, Bukhari IH, Rana UA (2017) A facile synthesis of new 5-aryl-thiophenes bearing sulfonamide moiety via Pd(0)-catalyzed Suzuki–Miyaura cross coupling reactions and 5-bromothiophene-2-acetamide: as potent urease inhibitor, antibacterial agent and hemolytically active compounds. J Saudi Chem Soc 21:S403–S414
Myrach T, Zhu A, Witte C-P (2017) The assembly of the plant urease activation complex and the essential role of the urease accessory protein G (UreG) in delivery of nickel to urease. J Biol Chem 292:14556–14565
Mobley H, Island MD, Hausinger RP (1995) Molecular biology of microbial ureases. Microbiol Rev 59:451–480
Khan KM, Wadood A, Ali M, Ul-Haq Z, Lodhi MA, Khan M, Perveen S, Choudhary MI (2010) Identification of potent urease inhibitors via ligand-and structure-based virtual screening and in vitro assays. J Mol Graph Model 28:792–798
Ibrar A, Khan I, Abbas N (2013) Structurally diversified heterocycles and related privileged scaffolds as potential urease inhibitors: a brief overview. Arch Pharm (Weinheim) 346:423–446
Gull Y, Rasool N, Noreen M, Nasim F-U-H, Yaqoob A, Kousar S, Rashid U, Bukhari I, Zubair M, Islam M (2013) Efficient synthesis of 2-amino-6-arylbenzothiazoles via Pd(0) Suzuki cross coupling reactions: potent urease enzyme inhibition and nitric oxide scavenging activities of the products. Molecules 18:8845
Amtul Z, Rasheed M, Choudhary MI, Rosanna S, Khan KM (2004) Kinetics of novel competitive inhibitors of urease enzymes by a focused library of oxadiazoles/thiadiazoles and triazoles. Biochem Biophys Res Commun 319:1053–1063
Frisch M, Trucks G, Schlegel HB, Scuseria G, Robb M, Cheeseman J, Scalmani G, Barone V, Mennucci B, Petersson G (2009) Gaussian 09, revision A, 2nd edn. Gaussian Inc., Wallingford, p 200
Roy D, Todd K, John M (2009) Gauss view; version 5. Semichem. Inc., Shawnee Mission
Bendary E, Francis R, Ali H, Sarwat M, El Hady S (2013) Antioxidant and structure–activity relationships (SARs) of some phenolic and anilines compounds. Ann Agric Sci 58:173–181
Serwar M, Akhtar T, Hameed S, Khan KM (2009) Synthesis, urease inhibition and antimicrobial activities of some chiral 5-aryl-4-(1-phenylpropyl)-2H-1,2,4-triazole-3 (4H)-thiones. Arkivoc 7:210–221
Pervez H, Ramzan M, Yaqub M, Nasim FuH, Mohammed Khan K (2012) Synthesis and biological evaluation of some new N4-aryl substituted 5-chloroisatin-3-thiosemicarbazones. Med Chem 8:505–514
Rehman AU, Rehman AU, Abbasi M, Khalid H, Dar P, Khan K (2012) Synthesis and biological screening of N-substituted derivatives of N-benzyl-4-chlorobenzenesulfonamide. Asian J Pharm Health Sci 2:384
Authors’ contributions
KR, MZ, NR, FUHN, SMB, made a significant contribution to experimental lab work, analysis and drafting of the manuscript. MR, VUA contributed for analysis of data, NBA and MNA, MNMA contributed to interpretation of data. TM and KA contributed towards computational studies. All authors read and approved the final manuscript.
Acknowledgements
The current study is the part of Ph.D. thesis research of Komal Rizwan. The Authors highly acknowledge Higher Education Commission (HEC), Pakistan, for providing scholarship (PIN No. 112-24510-2PS1-388) to Komal Rizwan. The authors also gratefully acknowledge the financial support by HEC (HEC Project No. 20-1465/R&D/09/5458). We are grateful to the Universiti Malaysia Pahang, Ministry of Education Malaysia FRGS (grant no. 150109).
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
All the main experimental and characterization data have been presented in the form of tables and figures. All the data is presented within the article.
Consent for publication
All authors consent to publication.
Ethics approval and consent to participate
Not applicable.
Funding
The research was funded by Higher Education Commission (HEC), Pakistan.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Authors and Affiliations
Corresponding authors
Additional file
Additional file 1: Figure S1.
HOMO/LUMO surfaces of compounds (3b–3p). Table S1. ESP values of compounds (3a–3p).
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Rizwan, K., Zubair, M., Rasool, N. et al. Palladium(0) catalyzed Suzuki cross-coupling reaction of 2,5-dibromo-3-methylthiophene: selectivity, characterization, DFT studies and their biological evaluations. Chemistry Central Journal 12, 49 (2018). https://doi.org/10.1186/s13065-018-0404-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13065-018-0404-7


